Swissmedic Certified - License No. 511593
Chronic care, built on the highest pharmaceutical
standards in the world.
SwissCore is a Swissmedic-certified pharmaceutical company that develops, delivers, and supports chronic treatments across 10 countries, with the same quality oversight as medicines sold in Switzerland, the EU, and the United States.
Swissmedic Certified
10 Countries
34 Own Medicines
Learn more:
One integrated ecosystem for chronic care.
Most pharmaceutical companies stop at the pill. SwissCore manages the entire treatment journey, from development through delivery, follow up, and renewal.
STEP 01

Develop
Our own medicines under Swissmedic standards. 150+ checkpoint audits.
STEP 02

Deliver
Direct to homes. Full cold-chain monitoring (35°C/3-hr excursion protocol)
STEP 03

Support
Reminders, guidance, and access to healthcare professionals.
STEP 04

Renew
Monthly renewal with correct execution and timing.
Swiss pharmaceutical quality. Verified at every step.
SwissCore holds an unlimited Swissmedic wholesale license (No. 511593-102616252) and GDP Certificate (GDP-CH-1001246). Swissmedic is Switzerland’s national authority — equivalent to the FDA and EMA.
Every batch undergoes triple independent lab testing in India, Costa Rica, and Switzerland before a Swiss Qualified Person authorizes release.
50+ checkpoints
22+ SOPs
3 countries lab testing
0 lots without QP approval
Swissmedic Licensed
FDA Equivalent
3 countries lab testing
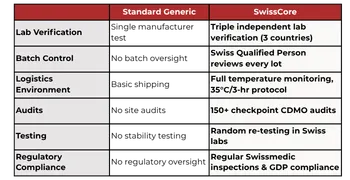
Our Unique Value Proposition:
Lab Verification
Batch Control
Logistics Environment
Audits
Testing
Regulatory Compliance
Standard Generic
Single manufacturer test
No batch oversight
Basic shipping
No site audits
No stability testing
No regulatory oversight
SwissCore
Triple independent lab verification
Swiss Qualified Person reviews every lot
Full temperature digital monitoring
150+ checkpoint CDMO audits
Random re-testing in Swiss labs
Regular Swissmedic inspections & GDP compliance
Our Unique Value Proposition:
Chronic treatments fail without structure
Without adherence and continuous support, chronic conditions become costly and difficult to control. We create the structure patients need to stay on track.
Who we work with.
We connect employers, insurers, providers, and patients through a single healthcare technological ecosystem focused on prevention, efficiency, and long-term outcomes.
What we deliver
Affordable Swiss-quality healthcare solutions focused on chronic disease management, adherence, prevention, and operational efficiency.

Let’s discuss how SwissCore can work for your organization.
